Compliance is not always about having the right documentation. In regulated industries, compliance is about proving that requirements are defined, changes are controlled, risks are addressed, tests are completed, and approvals are captured in a traceable, audit-ready way. When data is scattered across disconnected tools, that proof becomes difficult to maintain and defend during an audit. This is why traceability and auditability matter so much. Audit trails provide a time-stamped, tamper-resistant record of document access, edits, approvals, and transfers. They help organizations prove document integrity, data control, and accountability. In highly regulated environments, they are essential not only for passing audits but also for strengthening security.
For regulated organizations such as medical device, aerospace, and automotive manufacturers, the challenge is even greater due to product development spanning requirements, design, testing, quality, manufacturing, and change management. The most effective way to manage that complexity is through connected ALM and PLM systems that create a digital thread across the full lifecycle.
The Importance of Traceability and Auditability in Regulated Industries
Medical
For medical device manufacturers, traceability is foundational to both product safety and regulatory compliance. Teams must be able to demonstrate:
- How requirements were validated and verified
- How risks were managed
- How each component or process contributed to a safe and effective device
Without traceability, passing an audit or proving compliance becomes extremely difficult. It also becomes harder to manage recalls, prevent counterfeit products, and provide regulators with the clear evidence they expect. The stakes are high because failures can result in patient harm, financial losses, and serious damage to market trust. Additionally, medical device traceability supports stronger supply chain visibility. It does this by linking requirements to testing, risk management, and parts within the bill of materials.
That connection:
- Improves quality management
- Supports regulatory obligations such as UDI in the U.S. or EUDAMED in the European Union
- Helps manufacturers maintain the transparency required throughout the device lifecycle
Aerospace
In aerospace, traceability is critical because certification depends on continuous, evidence-based compliance. Programs must show how requirements were defined, implemented, verified, approved, and maintained across long development lifecycles. When requirements are treated as static documents instead of connected data, teams struggle to understand change impact, verification falls out of sync, and certification evidence becomes fragmented across tools. That often leads to rework, schedule delays, audit findings, and later entry into service.
Aerospace teams need to answer four questions at all times:
- What are we supposed to build?
- What changed and why?
- Where is the requirement implemented?
- What proof exists that it was verified and approved?
If those answers are incomplete or hard to retrieve, certification risk rises quickly. Strong requirements management supported by connected systems helps maintain bidirectional traceability between requirements, design, verification, and approvals. This makes compliance a continuous discipline rather than a last-minute scramble before an audit.
Automotive
In automotive, traceability is essential for safety, quality, compliance, and supply chain accountability. Manufacturers and suppliers need a complete record of each component’s journey, from raw material sourcing to final assembly. This allows them to quickly isolate affected parts, investigate issues, and take corrective action without disrupting the broader supply chain. This level of visibility is increasingly important as vehicles become more complex and OEMs demand stronger transparency across multi-tier supplier ecosystems.
Effective automotive traceability improves quality control, accelerates root cause analysis, limits recall scope, and supports regulatory and OEM compliance by automatically capturing and linking production and quality data. Instead of relying on incomplete manual records, manufacturers can use digital systems to create a verifiable trail that strengthens compliance posture and operational performance.
What Tools to Use: ALM and PLM Together
To achieve compliance success, organizations need systems that connect requirements, risks, tests, product structures, changes, approvals, and quality records across the full lifecycle. That is where ALM and PLM work best together.
ALM tools help organizations manage requirements, testing, risk, verification, and compliance activities. Meanwhile, PLM solutions connect product data, configurations, bills of materials, change processes, and lifecycle records across engineering and manufacturing. I n regulated environments, the value comes from linking these two worlds so that teams have end-to-end traceability instead of isolated data silos. In medical device development, for example, ALM integrated with PLM provides end-to-end traceability from design and development through manufacturing and post-market monitoring. In aerospace, connecting ALM and PLM supports synchronized hardware and software change management, audit-ready traceability, and a single source of truth for requirements and verification evidence.
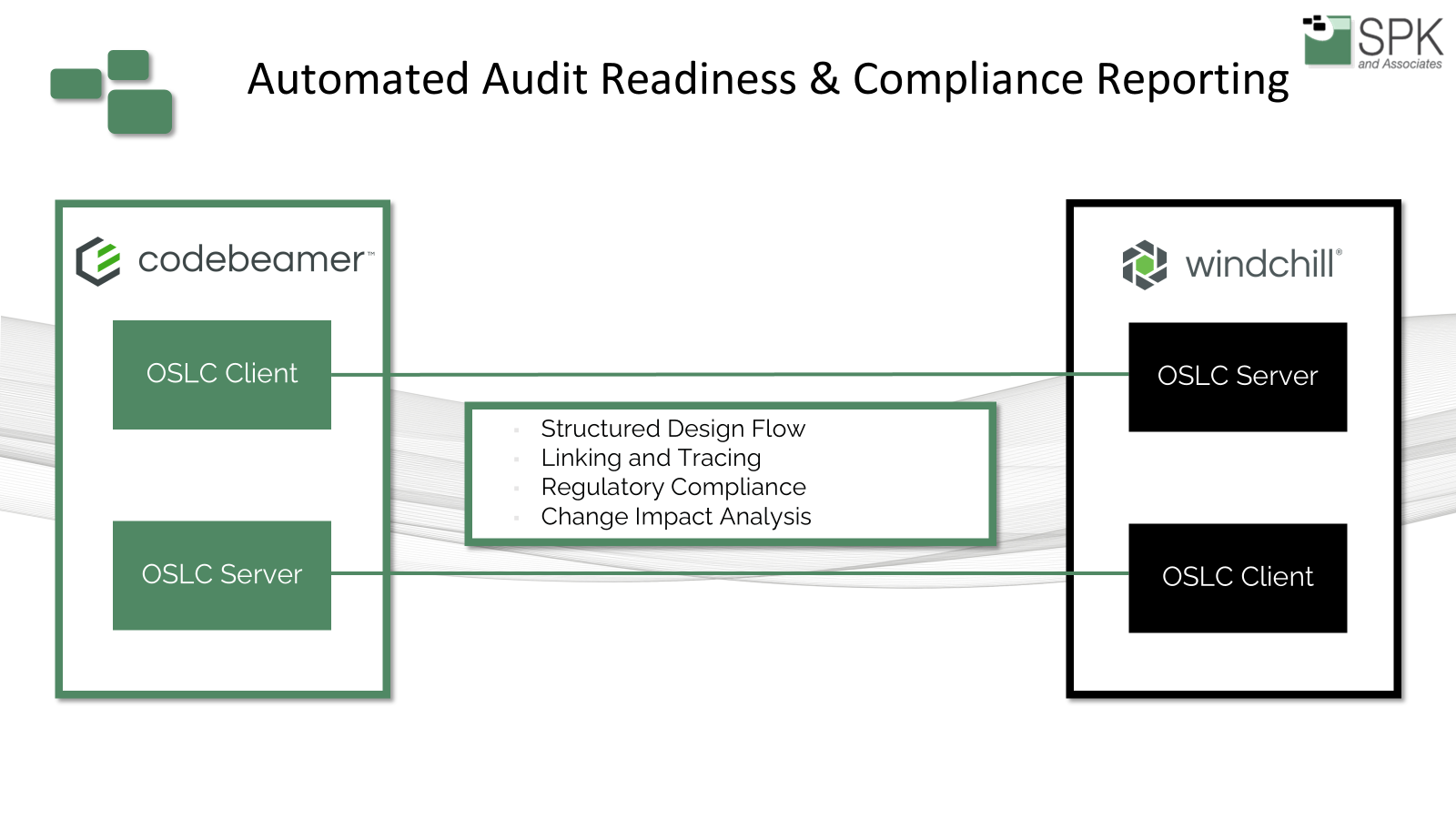
PTC Tools and the Digital Thread for Regulated Industries
Knowing which tools to utilize can be challenging. Our experts recommend PTC products, as they offer a strong foundation for this connected approach through Codebeamer and Windchill. Codebeamer provides ALM capabilities tailored for regulated industries. It helps teams manage requirements, risk, testing, and compliance with structured workflows and built-in support for regulated environments. Windchill extends that traceability into PLM by managing product data, changes, configurations, and related lifecycle information. Together, they create a unified digital thread that links intent, execution, and evidence across the product lifecycle.
This matters because compliance is much easier to sustain when all records are connected in real time. Rather than rebuilding evidence manually before audits, teams can maintain traceable relationships as part of daily work. That improves audit readiness, reduces compliance gaps, and gives organizations much greater confidence in their ability to manage change in a controlled manner.
Connecting the Systems You Already Use
While we recommend PTC solutions, we know not every organization starts with a fully unified PTC environment. Many companies already use a mix of ALM, PLM, quality, DevOps, ERP, or service management tools. In those environments, integration is still the key to compliance continuity.

That is where integration platforms like OpsHub can play an important role. A tool like OpsHub Integration Manager can connect the systems companies already rely on, helping synchronize requirements, defects, changes, and other lifecycle records across platforms. This helps reduce silos, preserve traceability, and support a more complete digital thread without forcing a rip-and-replace approach. For organizations that need to connect legacy tools, supplier systems, or separate engineering domains, this kind of integration can be a simple way to success.
Achieving Compliance Success with a Partner Like SPK
Compliance success requires more than software selection. It requires expertise in process design, traceability strategy, integration architecture, and regulated industry workflows. That is why working with an experienced partner matters. SPK helps organizations move beyond disconnected systems by aligning ALM, PLM, integrations, and cloud strategy around a complete digital thread. Whether a company needs to implement PTC tools, connect existing systems through integration, or improve hosting and operational management, SPK can help build an environment where traceability is continuous.
Our cloud hosting services help organizations run critical ALM and PLM platforms in environments designed for security, performance, and operational resilience. This means companies in the medical, aerospace, and automotive sectors can modernize their engineering ecosystem without taking on the complexity of managing it alone. A strong hosting strategy can support better uptime, easier administration, stronger control over sensitive engineering data, and a more scalable path toward connected compliance.
Achieving Compliance in Regulated Industries
In regulated industries, traceability and auditability are not optional. They are the foundation of compliance, quality, and accountability. In every case across medical device, automotive, and aerospace, disconnected tools make compliance harder than it should be. Connected ALM and PLM systems change that. By linking requirements, product data, risk, testing, change, and approvals into a unified digital thread, organizations can strengthen traceability, simplify audits, and respond to regulatory demands with far greater confidence. If you are ready to unlock simple compliance, reach out to our experts.